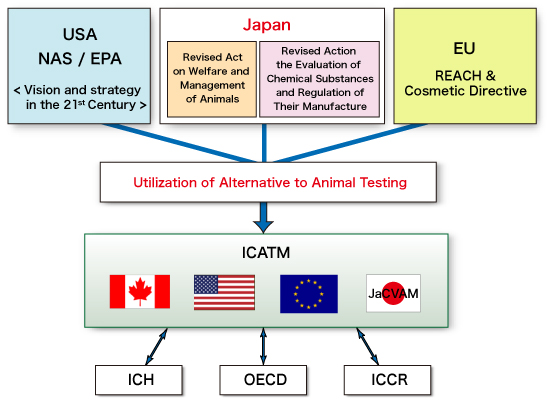
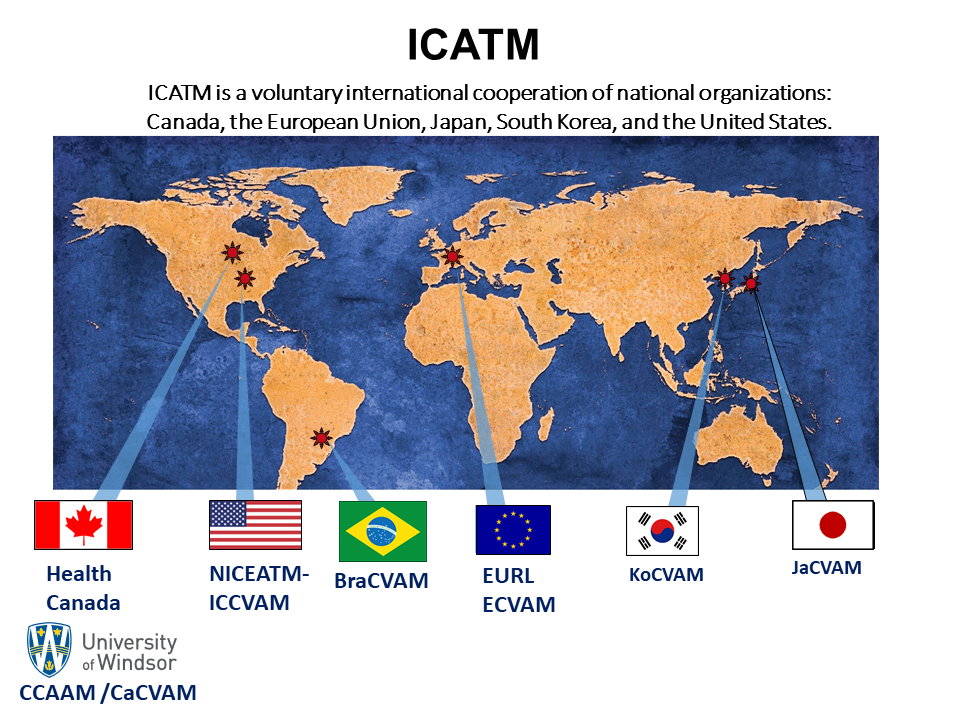
Information on ICATM
- Meeting documents
- 2023 EURL ECVAM Status Report – Non-animal Methods in Science and Regulation
- EURL ECVAM Recommendation on Non-Animal-Derived Antibodies (15MAY2020)
- Non-animal Methods in Science and Regulation (2020)
- Non-animal Methods in Science and Regulation(2021)
- A Strategic Roadmap for Establishing New Approaches to Evaluate the Safety of Chemicals and Medical Products in the United States
- 为评估美国化学品及医疗产品安全性建立新方案的战略发展蓝图
- 미국 내 화학 제품 및 의약품 안전 평가를 위한 새로운 접근법 확립을 위한 전략적 로드맵
- Um roteiro estratégico para o estabelecimento de novas abordagens de avaliação da segurança de produtos químicos e de produtos hospitalares nos Estados Unidos
- MEMORANDUM OF COOPERATION (pdf:161KB)
- MEMOIRE DE COOPERATION (pdf:185KB)
- NIEHS HP
http://www.niehs.nih.gov/news/releases/2009/pttw.cfm
ICATM meeting (remote) October 9, 2020
EURL ECVAM
NICEATM-ICCVAM
KoCVAM
Signing ceremony for the Modification to the ICATM Memorandum of Cooperation held at March 8, 2011
The Modification to the International Cooperation on Alternative Test Methods (ICATM) Memorandum of Cooperation (MOC) was signed in a ceremony March 8, 2011. The agreement expands the original ICATM MOC to bring the Republic of Korea into ICATM.


Stokes Helps Build Pacific Rim Partnerships for Advancing Alternative Testing (NIEHS News, January 2010)
Signing ceremony for the ICATM Memorandum of Cooperation held at April 27, 2009
Four country representatives signed the international agreement to reduce animal use in toxicity testing. Shown left to right are Linda Birnbaum, David Blakey, Elke Anklam, and Hajime Kojima representing Masahiro Nishijima of Japan. Standing are Marilyn Wind, Michelle Limoli of FDA, and William Stokes. (Photo courtesy of NIH) Birnbaum signed as the U.S. representative on behalf of the NTP Interagency.